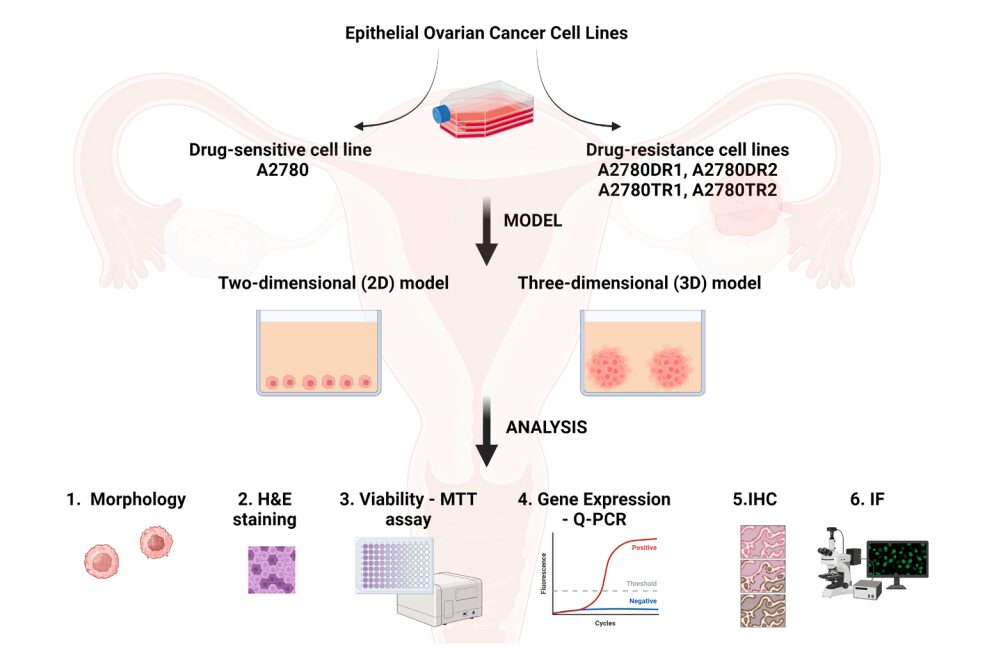
3D cell culture cancer models are increasingly recognized as essential tools to understand chemotherapy resistance and tumor behavior. In vitro cancer research has long relied on two-dimensional (2D) cell culture systems to study tumor biology and evaluate therapeutic candidates. While these monolayer-based models offer simplicity, reproducibility, and scalability, their physiological relevance remains limited.
A growing body of literature suggests that the structural organization of cells plays a decisive role in determining their behavior, particularly in response to treatment. A recent study focusing on ovarian cancer provides a compelling demonstration of this principle by comparing drug response, gene expression, and cellular adaptation in 2D versus three-dimensional (3D) culture systems.
The findings clearly indicate that cellular architecture is not a secondary parameter, but a central determinant of therapeutic resistance.
Experimental framework: comparing 2D and 3D tumor models
To investigate how spatial organization influences chemotherapy response, the study was designed to directly compare ovarian cancer cells cultured in two fundamentally different configurations.
Cells were exposed to two widely used chemotherapeutic agents with distinct mechanisms of action:
- Doxorubicin
- Topotecan
Importantly, the experimental setup included both drug-sensitive and drug-resistant cell lines, enabling the evaluation of resistance as both an intrinsic and adaptive phenomenon.
In conventional 2D conditions, cells grow as a monolayer, where exposure to nutrients, oxygen, and therapeutic compounds remains relatively uniform. In contrast, 3D cultures promote the formation of multicellular spheroids, in which cells self-organize into compact structures. This architecture leads to the emergence of spatial gradients and localized microenvironments, closely resembling those found in solid tumors.
Rather than being a simple variation in culture format, this shift introduces a controlled contrast between homogeneous and heterogeneous systems, enabling a precise assessment of how physical organization impacts cellular behavior and drug response.
To capture these effects comprehensively, the study integrates multiple levels of analysis, combining morphological characterization, viability assays, and gene expression profiling. This multi-parametric approach provides a robust framework for linking structural organization to functional outcomes.
The overall experimental workflow is summarized below.
Figure 1. Experimental design comparing chemotherapy response in 2D and 3D ovarian cancer models; Drug-sensitive and drug-resistant ovarian cancer cell lines were cultured in 2D monolayers and 3D multicellular spheroids. Cellular responses were evaluated through morphology, viability, and gene expression analyses, highlighting the impact of spatial organization and microenvironment on drug resistance.
A profound increase in chemoresistance in 3D systems
One of the most striking outcomes of the study is the consistent observation that cells cultured in 3D exhibit significantly enhanced resistance to chemotherapy.
Compared to 2D cultures, spheroid-based systems show:
- reduced drug sensitivity across multiple cell lines
- increased survival following exposure to cytotoxic agents
- persistence of viable subpopulations even at higher drug concentrations
Importantly, this phenomenon is not restricted to intrinsically resistant cells. Even cell lines initially categorized as drug-sensitive demonstrate a marked decrease in responsiveness when transitioning to a 3D configuration.
This result suggests that resistance can emerge as an adaptive response to the microenvironment, rather than being solely driven by genetic mutations.
Microenvironmental complexity as a driver of resistance
The increased resistance observed in 3D cultures is closely linked to the emergence of a tumor-like microenvironment. Unlike 2D systems, where conditions are largely homogeneous, 3D spheroids generate spatial heterogeneity that profoundly influences cellular behavior.
Within these structures, several key features develop simultaneously:
- oxygen gradients leading to hypoxic cores
- limited diffusion of nutrients and metabolites
- restricted penetration of therapeutic agents
- intensified cell–cell interactions and signaling
These parameters recreate essential aspects of in vivo tumors, providing a more realistic context for studying treatment response.
Key mechanisms underlying resistance in 3D models
The study identifies a combination of physical and biological mechanisms that contribute to the enhanced resistance observed in spheroids.
Among the most relevant processes are:
- Impaired drug penetration
The outer layers of the spheroid act as a barrier, limiting the diffusion of chemotherapeutic agents toward the core - Hypoxia-driven adaptation
Reduced oxygen levels activate survival pathways and promote metabolic flexibility - Enhanced intercellular communication
Close proximity between cells facilitates signaling networks that support collective resistance - Heterogeneous exposure to stress
Variations in nutrient and drug availability lead to the coexistence of multiple cellular states within the same structure
These mechanisms do not operate independently but rather interact to produce a robust and multifactorial resistance phenotype.
Gene expression reprogramming induced by 3D organization
Beyond structural constraints, the transition to a 3D environment triggers significant changes at the molecular level. Gene expression analyses reveal a coordinated activation of pathways associated with survival and resistance.
In particular, the study reports:
- increased expression of drug efflux transporters, reducing intracellular drug accumulation
- activation of anti-apoptotic pathways, allowing cells to withstand cytotoxic stress
- induction of hypoxia-responsive genes, reflecting adaptation to oxygen gradients
This demonstrates that the 3D context does not merely protect cells physically but actively induces a functional reprogramming of their phenotype.
Spatial heterogeneity and its impact on therapeutic response
A defining characteristic of 3D tumor models is the emergence of spatial heterogeneity. Within a spheroid, cells are exposed to different microenvironments depending on their position.
Cells located at the periphery experience higher drug concentrations and better oxygenation, while those in the core are relatively shielded. This results in the formation of distinct subpopulations with varying sensitivities to treatment.
Such heterogeneity is absent in 2D cultures, where all cells are uniformly exposed. Consequently, 2D systems fail to capture a critical dimension of tumor biology: the coexistence of resistant and sensitive cells within the same structure.
Implications for drug development and preclinical evaluation
The findings of this study have direct implications for the design of preclinical models. They highlight the limitations of conventional 2D systems and underscore the necessity of integrating 3D approaches into drug screening pipelines.
More specifically, the results suggest that:
- drug efficacy may be systematically overestimated in 2D models
- resistance mechanisms can remain undetected without spatial context
- microenvironmental factors must be considered in therapeutic evaluation
As a result, transitioning toward 3D systems is not simply an improvement in model complexity, but a requirement for biological accuracy and translational relevance.
Toward controlled and reproducible 3D systems
Despite their advantages, current 3D models still face significant challenges. Variability in spheroid size, lack of control over internal gradients, and limited reproducibility can compromise experimental reliability.
The next generation of in vitro systems must therefore aim to combine:
- the biological relevance of 3D architecture
- precise control of microenvironmental parameters
- scalability and reproducibility compatible with industrial applications
Achieving this balance will be essential to fully unlock the potential of 3D cell culture in oncology.
Conclusion
This study provides a clear and compelling demonstration that three-dimensional cellular organization fundamentally alters chemotherapy response in ovarian cancer.
By revealing resistance mechanisms that remain hidden in traditional 2D models, it underscores the importance of incorporating spatial and microenvironmental complexity into preclinical research.
As the field moves toward more predictive and physiologically relevant models, the ability to accurately reproduce and control the tumor microenvironment will become a defining factor in the development of effective therapies.