For many years, cellular mechanics was primarily studied in two-dimensional systems. Cells were plated on flat elastic substrates, and their traction forces were quantified with high precision. These models revealed fundamental principles: actomyosin contractility drives force generation, focal adhesions transmit stress, and substrate stiffness regulates cellular behavior.
However, tumors do not develop on flat surfaces. They evolve within three-dimensional extracellular matrices where fiber organization, confinement, and local heterogeneity reshape mechanical interactions. In this context, force generation cannot be understood as a purely intrinsic cellular property. It emerges from continuous mechanical reciprocity between cells and their microenvironment.
Recent advances using deformable hydrogel microspheres now enable direct quantification of cellular stresses inside collagen-embedded tumoroids. By combining high-resolution surface reconstruction with single-cell segmentation, it becomes possible to map traction fields in three dimensions and assign mechanical signatures to individual cells.
Three-Dimensional Stress Sensing at the Cellular Scale
Deformable hydrogel microparticles, comparable in size to individual cells, function as embedded mechanical probes. Unlike collagen, whose nonlinear and heterogeneous mechanics complicate modeling, these particles possess defined and tunable elastic properties. Their deformation directly reflects stresses transmitted by neighboring cells.
High-resolution confocal imaging enables reconstruction of particle surfaces with nanometric precision. Sub-micrometer deformations can be detected and translated into spatially resolved traction fields with sensitivities in the tens of pascals range. Measured stresses typically range between 10 and 100 Pa, corresponding to integrated forces from 0.1 to 100 nN.
These values align with physiological force magnitudes previously reported in two-dimensional systems. However, the three-dimensional context reveals additional mechanical complexity driven by spatial constraints and matrix architecture.
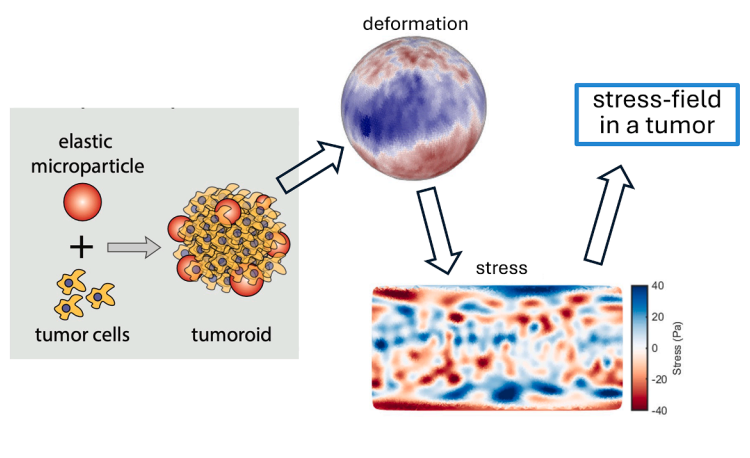
Figure – Conceptual overview of deformable microparticle–based traction mapping in 3D tumoroids.
An elastic hydrogel microparticle is embedded within a collagen-embedded tumoroid. Cellular forces deform the particle surface. High-resolution reconstruction of particle deformation enables computation of spatially resolved stress fields at the particle boundary, allowing mapping of mechanical stresses within the tumor microenvironment.
Single-Cell Force Attribution Within Tumoroids
A critical innovation lies in coupling particle deformation analysis with three-dimensional cell segmentation. By labeling nuclei and cytoskeletal actin, individual cells inside the tumoroid can be computationally separated and spatially mapped relative to the embedded microsphere.
This strategy enables decomposition of traction fields into cell-specific contributions. Instead of reporting averaged stress distributions, it becomes possible to determine how much force each cell exerts, over which contact area, and in which direction.
Mechanical heterogeneity becomes immediately apparent. Adjacent cells within the same tumoroid can generate markedly different force magnitudes. Tumor mechanics is therefore intrinsically heterogeneous rather than uniform.
Contact Area as a Determinant of Force Transmission
One of the most robust observations is the relationship between interaction area and net force. Cells that establish larger contact regions with the deformable probe generate proportionally greater integrated forces.
This scaling behavior resembles predictions from classical contact mechanics, particularly Hertzian models describing interactions between soft elastic bodies. Although living cells are neither homogeneous nor isotropic, their effective mechanical behavior at micrometer scales approximates continuum elasticity principles.
This convergence suggests that collective cytoskeletal forces average into predictable mechanical patterns despite molecular complexity.
Actin Organization and Mechanical Output
Cytoskeletal architecture remains central to force generation. Regions enriched in cortical actin often correlate with increased local traction. However, this relationship is not strictly linear. Variability between cells indicates that actin abundance alone does not fully determine mechanical output.
Force transmission efficiency depends on effective coupling between the cytoskeleton, adhesion complexes, and extracellular resistance. In three-dimensional collagen matrices, this coupling is modulated by fiber alignment, pore size, and local compliance.
Mechanical behavior therefore reflects both intracellular contractility and extracellular structural context.
Mechanical Heterogeneity Within the Tumor Microenvironment
Unlike uniform two-dimensional substrates, collagen matrices exhibit structural anisotropy and spatial heterogeneity. Cells encounter variable mechanical resistance depending on their local niche.
Embedded microsphere sensing reveals that cells integrate these environmental constraints into their force generation strategies. Some cells primarily exert compressive stresses, while others produce distributed traction patterns mediated by matrix fibers. The resulting force landscape is spatially structured rather than radially symmetric.
Interestingly, global cell morphology does not reliably predict traction magnitude in three dimensions. Mechanical output appears to depend more strongly on local matrix interactions than on overall cell elongation.
Implications for Tumor Progression and Therapy
Mechanical forces influence migration, extracellular matrix remodeling, mechanotransduction pathways, and therapy resistance. Quantifying these forces directly within tumoroids bridges the gap between reductionist mechanical assays and physiologically relevant tumor models.
This methodology enables comparison of mechanical phenotypes across tumor subpopulations and assessment of how therapeutic interventions alter force transmission. It provides a platform to investigate whether mechanical signatures correlate with invasive potential or treatment response.
By resolving cellular stresses at micrometer scales inside three-dimensional microenvironments, tumor mechanics becomes a measurable parameter rather than an inferred concept.
Conclusion
Tumoroids are mechanically active systems in which each cell continuously interacts with its surroundings. Deformable microsphere-based traction mapping demonstrates that cellular forces in three-dimensional matrices are spatially organized, heterogeneous, and strongly influenced by local contact architecture.
Understanding cancer progression therefore requires integrating molecular profiling with spatially resolved mechanical characterization. Quantifying forces where they are generated adds a critical dimension to tumor biology, linking structure, mechanics, and cellular behavior within a unified framework.
Scientific background: Deformable hydrospheres and cell segmentation report on stresses generated by individual cells in tumoroids, Cell Reports Physical Science, 2026.