Beyond biochemistry: a new layer of genomic regulation
For decades, epigenetics has been defined as the set of reversible modifications that regulate gene expression without altering DNA sequence. DNA methylation, histone modifications, and chromatin remodeling have long been considered the principal mechanisms governing cellular differentiation and lineage stability.
However, a major conceptual shift is now emerging in molecular cell biology. Physical forces themselves participate directly in genomic regulation. Mechanical constraints are no longer viewed as passive environmental conditions. They are increasingly recognized as structural regulators of nuclear organization and chromatin architecture.
This emerging field can be described as mechano epigenetics, as it reveals that physical signals are capable of inducing stable changes in chromatin structure and influencing gene expression in a lasting manner.
The nucleus as a mechanical sensor
Recent work in nuclear mechanotransduction has demonstrated that the nucleus is not a passive organelle insulated from extracellular forces. It is physically connected to the cytoskeleton through specialized protein complexes that allow mechanical signals to propagate from the cell membrane to the nuclear interior.
When a cell experiences shear stress, stretching, compression, or geometric confinement, these forces are transmitted through the cytoskeletal network to the nuclear envelope. This mechanical coupling modifies nuclear tension and alters the spatial organization of chromatin.
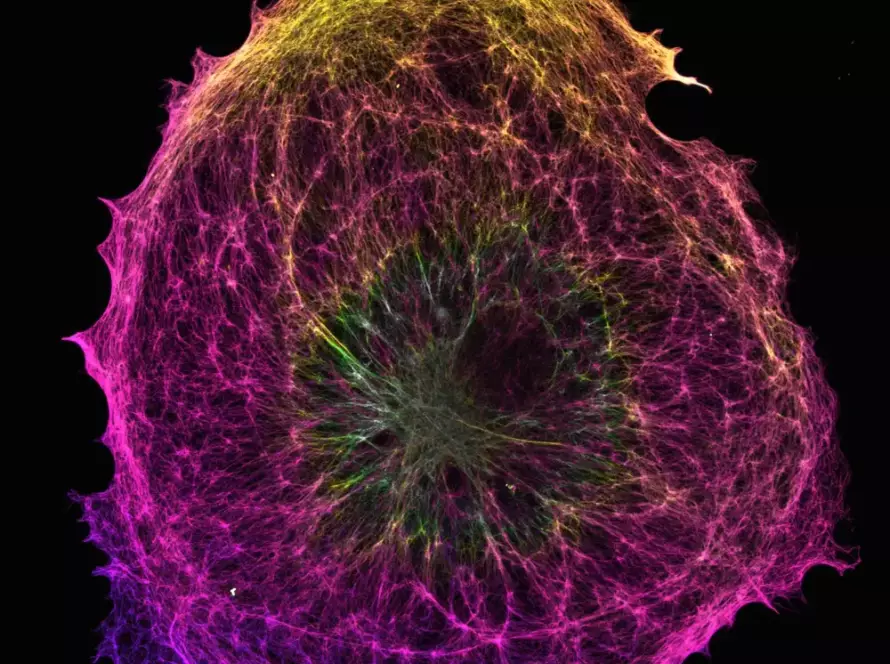
The three dimensional architecture of the genome becomes responsive to force. Chromatin can reorganize in response to deformation, modifying accessibility to transcriptional machinery and influencing gene expression profiles.
This phenomenon is not purely transient. Mechanical forces can induce durable alterations in chromatin configuration, thereby shaping functional genomic states.
Force dependent chromatin remodeling
Chromatin is not a rigid scaffold. It exhibits viscoelastic properties that allow it to respond dynamically to mechanical deformation. Experimental studies have shown that direct mechanical stretching of chromatin can promote transcriptional activation by increasing local accessibility of genomic regions.
Mechanical stress can influence histone modifications, chromatin compaction, and the redistribution of heterochromatin domains. In this context, the mechanical environment becomes a determinant of genome organization.
This reframes our understanding of gene regulation. Biochemical signaling pathways remain fundamental, but they are complemented by a mechanical dimension of epigenetic control. The physical state of the cell directly influences genomic structure and function.
Cellular mechanical memory
One of the most intriguing aspects of mechano epigenetics is the concept of mechanical memory. Cells exposed to defined mechanical environments can retain transcriptional signatures even after the original stimulus is removed.
For example, cells cultured on rigid substrates may preserve altered gene expression patterns following transfer to softer environments. This suggests that mechanical experience leaves a persistent imprint on chromatin architecture.
Mechanical memory does not involve genetic mutation. It reflects stabilization of epigenetic states induced by physical constraints. These stabilized states can influence subsequent cellular responses and differentiation trajectories.
This concept has profound implications for developmental biology, disease modeling, and biomanufacturing.
Toward intergenerational transmission of mechanical states
If mechanically induced chromatin states are sufficiently stabilized, they can be propagated through cell division. While the extent of transgenerational inheritance in mammals remains under investigation, the possibility that physical forces influence long term lineage programming is gaining attention.
In tissues subjected to chronic mechanical stress such as vascular systems, musculoskeletal structures, or tumor microenvironments, cells may integrate mechanical cues into durable epigenetic programs.
The boundary between physical environment and cellular identity becomes less rigid than previously assumed. Mechanical context may contribute to lineage specification, disease progression, and therapeutic responsiveness.
Implications for three dimensional cell culture and biomanufacturing
The recognition that physical forces influence epigenetic states carries direct implications for advanced cell culture systems.
If shear stress, flow dynamics, and confinement influence chromatin organization, then the mechanical environment of a bioreactor becomes a decisive variable. Excessive shear or heterogeneous mechanical conditions may alter genomic stability, differentiation potential, and batch reproducibility.
Conversely, a controlled three dimensional culture environment with precisely regulated shear can serve as a platform for passive epigenetic engineering. By modulating physical constraints without introducing exogenous biochemical factors, it becomes possible to influence transcriptional landscapes in a stable and reproducible manner.
This perspective transforms how we view mechanical control in biomanufacturing. Shear regulation is not merely protective. It becomes strategic. Mechanical stability may preserve cellular integrity, maintain stemness, and reduce unwanted phenotypic drift.
In this framework, low shear three dimensional systems offer more than gentle mixing. They provide an opportunity to align mechanical conditions with genomic stability.
A new biological frontier
Mechano epigenetics expands our understanding of how cells interpret their environment. The genome is not regulated solely by molecular signaling cascades. It is also shaped by physical laws.
As regenerative medicine, organoid technologies, and advanced cell therapies continue to evolve, the ability to integrate mechanical control into epigenetic stability may become a defining factor in scalable and reliable bioproduction.
The genome is not written only by chemistry. It is also sculpted by physics.